 Let's do some examples, how about cobalt? Well cobalt's full electron configuration is argon 4s2 3d7, but remember the electrons that are symbolized by argon and brackets are the core electrons. So some people would say that those aren't really valence electrons, but I'm going to say the outermost d-sub-level, we're going to call valence electrons for all the transition metals. As it turns out for zinc, the d electrons are not used for bonding. 
Zinc would have the full electron configuration given by argon 4s23d10. So that's what copper does, it promotes one of it's s electrons into the d sub-level so that it can have a filled d shell. Remember when n gets high, when n gets up to four, five and six, the energy levels are very, very close together anyway, do you remember that from the Bohr model of the hydrogen atom? So it doesn't take much energy to change the electron from one atom to another once we're that far away from the nucleus. So sometimes the transition metals will promote one or even two of the s electrons into the d sub-level so that the d sub-level is half filled or fully filled. Copper does something a little bit strange, the transition metals, it turns out, likes to have their d sub-level either half filled or fully filled. And there are eight electrons in those high energy d orbitals. If we were going to ask, how many valence electrons does nickel have, the answer would be ten, because there's two electrons in the highest energy s orbital. But in the transition metals, sometimes the d electrons are very reactive, so, for example, for nickel. Now, if we went back to the previous slide, we were not counting the d, any electrons that are in the d sub-level, we were not counting those as valence electrons for the main group elements, were we? For the main group elements, we were considering the d electrons as part of the core electron set. The issue gets slightly more complicated for transition metals, in transition metals the outermost or highest energy s electrons are still considered to be valence electrons because we do observe reactions where the s electrons are involved, and usually we count all of the outermost d electrons. Tin is in group 14, so it has four valence electrons, how about selenium? Here's one for you to try, how many valence electrons does selenium have? Thank you for answering, if you look on the periodic table, you see that selenium is in group sixteen, which means selenium has six valence electrons. So, you can pick any of the main group elements and determine how many valence electrons it has, for example, tin. And over here at 18 we would have the noble gases. Then we would skip the transition metals, which is ten columns worth and we end up with 13, 14, 15, 16, and 17 for these group numbers. So here I have some old numbering of the group numbers, remember now we use just numbers from one to 18, if we did that, we'd have one and two for the first two columns. It doesn't matter which period the element's in, if it's in one of the main groups, so we're not counting the transition metals or the inner transition metals, we can look at the group number to determine how many valance electrons are present. And all electrons in partially filled sub-levels, so, for example, a sodium atom, which we just looked at, has one valence electron and that valence electron is in the 3s orbital. For the main group elements, the valence electrons are the highest energy s electrons. Valence electrons are very easy to count for the main group elements. That's the electron that leaves the sodium when the neutral sodium becomes sodium cation. Sodium has 1 valence electron, which is in a 3s orbital. There are 10 electrons in neon, and those are all core electrons of sodium. So, for example, if I had a sodium atom, I can write the electron configuration using the noble gas core of radiation, and the core electrons are summarized with just this symbol for neon in brackets. And it's the outermost valence electrons that leave the atom if it becomes a cation. It's those outermost valence electrons that are involved in chemical bonding. 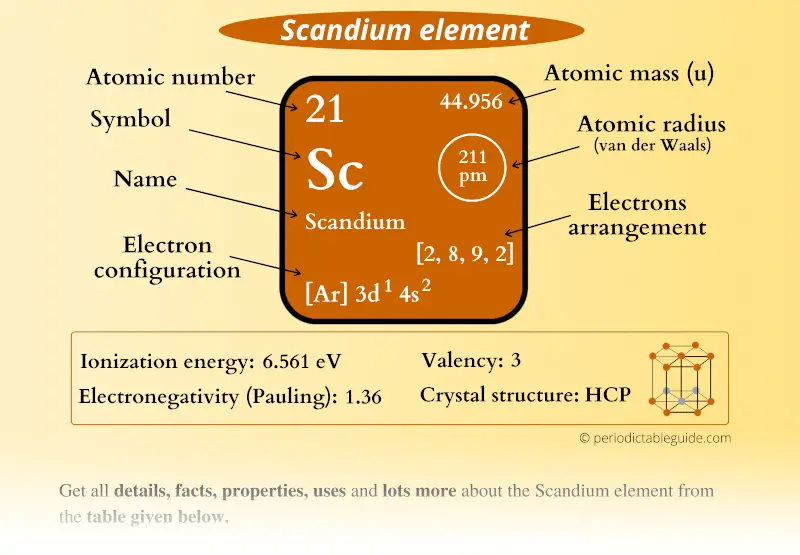
Those are the ones I've been considering when we talked about properties like ionization energy. The valence electrons are the outer most electrons. 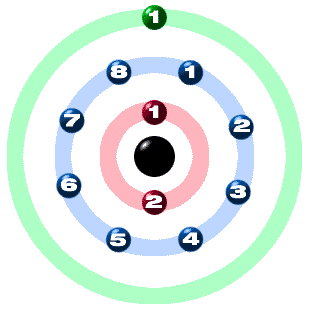
The core electrons are those bound inner electrons that are at the lower energy level, that are not involved in chemical reactions. Electrons can be divided into two types, the core electrons, and valence electrons. We did that, for example, when we were calculating formal charge in some of the molecules that I've drawn out. I know that, throughout the course, I've already mentioned, sometimes, the word, valence electron. The last thing I'd like to talk about is different types of electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
